Chemistry
Stoichiometry
Concept of relative atomic masses, molecular masses, and molecular formulae.
Calculation of relative atomic masses, relative molecular masses considering existence of isotopes, understanding empirical formula, molecular formula, mass and formula mass of different compounds.
Validation of law of stoichiometric proportions to introduce concept of moles. Includes mole-mole, mole-mass, mass-mass, mass-volume conversions.
Application of concept of moles to work out various stoichiometric proportions like % yield, % purity, reacting masses of reactants and products, limiting reagents, atom economy.
Extending the concept of mole to understand gaseous reactions and calculation of concentration of solutions through measuring volumes of solutions required by titration method, concentration numericals.
What is Stoichiometry?
Stoichiometry bridges the gap between macro quantities like mass and micro quantities like atoms and molecules.
Laws of Stoichiometry:
- Law of Conservation of Mass
- Law of Constant Proportions
- Law of Multiple Proportions
- Law of Reciprocal Proportions
- Law of Gaseous Volume
Moles
A mole is a fixed constant in chemistry. Similar to how one dozen equals twelve objects, one mole equals 6.02 × 1023 atoms of an element or compound.
Number of atoms in 1 mole of gold = 6.02 × 1023
Number of atoms in 1 mole of hydrogen = Number of atoms in 1 mole of gold
The atomic weight/relative atomic mass of an element is equal to its molar mass in grams. The molar mass of an element is the mass of 1 mole of it (6.02 × 1023 atoms).
Example: The atomic weight/relative atomic mass, and thus molar mass in grams, of 1 mole of oxygen is 15.999 g/mol (~16.0 g/mol).
Two moles of oxygen = 16 × 2 = 32 g/mol.
Mole-Mole Relationships
In a balanced equation, the number of moles of a compound is given by the coefficient in front of it. By finding the ratio of the number of moles of reactants to products, it is possible to find the mass of products formed for a given mass of reactants.
Mole-Volume Relationships
1 mole of any gas at RTP (Room Temperature and Pressure)/SATP (Standard Ambient Temperature and Pressure) [298 K, 105 pascals] will occupy 24 dm3 of volume.
1 mole of any gas at STP (Standard Temperature and Pressure) [273 K, 105 pascals] occupies a volume of 22.7 dm3.
Also, if all the reactants are gases, then you can take the mole ratios as the volume ratios! Essentially, you don’t need to find the number of moles or the molar mass or anything like that, you can just directly take the coefficient as the volume of the gas in dm^3.
Molecular and Empirical Formula
The molecular formula is the normal formula of a compound.
For example, glucose’s molecular formula is C6H12O6.
The empirical formula is the simplest formula of a compound down to the simplest ratio.
Going back to glucose, its empirical formula is CH2O, simplifying the ratio of 6:12:6 to 1:2:1.
The empirical formula is a step in finding the molecular formula of a compound. First, the percentage composition of a compound is found, meaning what elements it is made of and how much of a percentage each element makes up. From there, we derive the empirical formula, where the ratio is then multiplied to find the molecular formula.
The steps to find the empirical formula are as follows:
- Find out the masses (in grams) of each element present.
- Find the number of moles of each element.
- Find the ratio of the moles of each element to each other.
- Based on the ratio, write the formula.
When the percentage composition of an element is given, !!assume that if an element was 100% present, its given mass would equal 100g. So if a compound is 60% carbon, there is 60g of carbon in there.
Limiting Reagents:
If reagents aren’t added in appropriate ratios, and there isn’t enough of one reactant, it limits the product yield. The amount of product formed always depends on the limiting reagent.
First, find the ratio between the moles of the reactants. Then, based on the number of moles of the limiting factor, find the number of moles of the other reagent that can react with it, and then the ratio of the product moles to the limiting reagent moles. From there, find the mass of the product formed.
The reactant in short supply is limited.
The reactant that is not in short supply or more than required is excess.
% Composition:
Divide the molar mass of the element in the compound by the total molar mass of the compound and multiply it by a hundred to get the % composition, or how much of the compound is made of that element.
% Yield:
This refers to how much of the calculated mass was actually obtained, and is given by the formula of actual mass obtained/calculated mass to be obtained * 100/1.
% Purity:
Dividing the molar mass of the pure sample extracted from the total molar mass of the impure sample provides us the % purity of the compound, showing how much of it was made of the pure specified chemical substance.
Concentration:
The concentration of a solution is equal to the number of moles of the solute divided by the volume of the solvent, and is given by the formula:
Where:
- C = concentration (moles per decimeter cubed, a.k.a. mol/dm³)
- n = the number of moles of the solute
- V = the volume of the solvent in cm³ or dm³
Remember that 1 dm³ = 1000 cm³.
A Short Note on Acids and Bases
Any salt contains a metal cation and a nonmetal anion. NaCl (Sodium chloride) has sodium (Na) and chlorine (Cl).
In the formation of a salt, the metal cation always comes from a base, whereas the nonmetal anion comes from an acid.
Bases consist of metals and their various compounds, namely metal oxides, metal hydroxides, metal carbonates, and metal hydrogen carbonates.
There are three main acids to consider as of now: H2SO4 (sulfuric acid), HNO3 (Nitric acid), and HCl (hydrochloric acid).
When an acid and base of the right pH and in the right concentration react, they neutralize each other or essentially “cancel” each other out, almost always forming water and a metal salt along with releasing a gas of some sort.
When an acid and base react and don’t form water, then no neutralization has taken place, and only a redox reaction has occurred.
Examples of Acid-Base Reactions:
| Acid | Base | Product |
|---|---|---|
| HCl | Na | NaCl + H2 (No water, so redox, not neutralization!) |
| HCl | Na2O | NaCl + H2O |
| HCl | NaOH | NaCl + H2O |
| HCl | Na2CO3 | NaCl + CO2 + H2O |
| HCl | NaHCO3 | NaCl + CO2 + H2O |
Acids and bases must be of the right concentration and volume to neutralize each other.
Finding the number of atoms in a covalent compound:
Multiply the number of moles of the compound (the coefficient) with Avogadro’s constant (6.02 x 10^23) to get the number of molecules of that compound.
Because 1 mole of that compound will have 6.02 x 10^23 molecules of it. Hence, considering cross multiplication, another number of moles will have that number of moles times 6.02 x 10^23.
For the number of atoms of a certain element, multiply the number of atoms of that element in one molecule of the compound with the coefficient with Avogadro’s constant.
For the total number of atoms, do the same as above. Multiply the total number of atoms in 1 molecule of the compound with the coefficient with Avogadro’s constant.
For ionic compounds, just write formula units because ionic compounds can’t be split into atoms!
% Atom Economy:
The % atom economy is a measure of the ratio of the masses of useful products from the chemical reaction to the total masses of products from the reaction. If there is only a single product produced in a reaction, then 100% atom economy is reached!
Green Chemistry
This is a branch of science that focuses on reducing both the usage and production of hazardous and harmful substances in chemistry. This could be by swapping out a harmful chemical for a non-harmful one that does the same job.
They also aim to do this by reducing any harmful waste produced through a reaction, such as through boosting the % atom economy – the greater the mass of the useful product formed, the less the mass of the waste produced.
It also aims to reduce the amount of nonrenewable resources used within chemical reactions (like fossil fuels) by making reactions more efficient and thus not needing to be repeated, by once again increasing the % atom economy.
12 Principles of Green Chemistry
- Maximize atom economy: By ensuring that the mass of useful products is the greatest, this means that the majority of the masses of the reactants is going into useful products, meaning little to no atoms are “wasted”.
- Minimize the potential for accidents: Self-explanatory. By only utilizing or promoting reactions where any and all forms of the reactants and products have the least potential to cause harm, the principles of green chemistry are upheld.
- Design safer chemicals and products.
- Analyze in real-time to prevent pollution: To reduce the formation of unnecessary and undesirable byproducts that are a waste of reagents, monitor the reaction as it takes place.
Atomic Mass & Relative Atomic Mass
The mass of a single atom of lithium in amu is equal to the mass of 1 mole of lithium in grams; because Avogadro's constant (which is 6.02 × 10^23).
Reasoning behind Relative Atomic Mass: Dalton said fuck no ain’t no way we’re ever finding how heavy a single teeny tiny atom is.
Instead he found that the mass ratios of hydrogen and oxygen when reacting in big, measurable quantities of grams in the lab, was equal to 1:16. And he found that for whatever mass of hydrogen and oxygen he took, the mass ratios would be the same.
And since according to Avogadro’s law all gases at RTP have same number of molecules and same volume, it meant that the mass ratio of even 1 atom of hydrogen and 1 atom of oxygen was still 1:16.

“Equivalent numbers of atoms”
https://youtu.be/TSQlM72_MiU?si=AWW6WFS98U0GiknN
 Because the mass ratios are the same across all sizes, 1 atom of oxygen has a relative atomic mass of 16 a.m.u. and 1 mole of oxygen has a molar mass of 16 g/mol.
Because the mass ratios are the same across all sizes, 1 atom of oxygen has a relative atomic mass of 16 a.m.u. and 1 mole of oxygen has a molar mass of 16 g/mol.
It’s like g/ml and kg/L. The mass ratio is the same because both units are being increased by the same factor of x 1000.
But for chemistry, that factor is 6.02 × 10^23 (Avogadro’s constant).
Titration
This is the process of neutralizing an acid with an appropriate base to form salt and water, which is an aqueous solution. For example:
- Measure out 10cm³ of NaOH to a conical flask, using a pipette for maximum accuracy.
- Add a few drops of the indicator to the flask.
- Swirl the flask till the two substances mix and turn a dark pink.
- Gradually add drops of the HCl to the flask, swirling the flask to ensure even mixing, till the solution turns clear and colourless. At that point, it has been completely neutralized.
- Measure the volume of HCl used to neutralize 10cm³ of NaOH by looking at the lower meniscus; that is to say, if the liquid in the tube is curved, then look at the lowermost part of the curve for the measurement.
- Evaporate the salt to remove water, leaving behind impure salt.
- Add coke or another substance to absorb the indicator in the salt, leaving behind pure salt.
The acid in this case is considered to be neutralized when the phenolphthalein added to the base using a pipette turns from pink to colourless.
Titration can be used to calculate the concentration of an unknown solution based on how much base or acid is used to neutralize it.
Unit 2: Impacts of Chemical Industries
Theories of Acids and Bases - Bronsted-Lowry, Arrhenius theory, autoionisation of water, ionic product of water, understanding of development of pH scale based on Hydrogen and hydroxide ion concentration.
Physical properties of acids and bases.
Strength of acids & bases - understanding weak and strong acids on basis of dissociation.
pH scale and numericals.
Preparation of salts – method of preparation of soluble salts and insoluble salts including acid-base titrations.
Real life applications of acids and bases.
Chemical properties of acids - reaction with bases, metals, metal carbonates, metal oxides.
Chemical properties of bases – reaction with acids and ammonium salts.
Salt Analysis: Solubility of salts, Test for anions [Carbonate, nitrate, chloride, bromide, iodide, sulphate, sulphite], cations [Ammonium, aluminium, iron (II), Iron (III), Copper (II), Chromium, zinc, calcium] and gases [oxygen, ammonia, chlorine, hydrogen, carbon dioxide].
Hardness of water and its effects.
Acids and Bases
In terms of physical properties…
Acids are sour to taste (citric acid), corrosive (hydrochloric acid) and show low pH values (2,3,4, etc.)
Bases are bitter (don’t try this at home, kids!), soapy to the touch (NaOH), slightly corrosive, and have high pH values (12,13,14, etc.) They do not react with metals because metals tend to be slightly basic themselves, unless they are amphoteric (contain both acidic and basic properties) and conduct electricity.
But these are not that accurate, because just because something tastes a certain way doesn’t mean it's acidic or basic.
Meanwhile, on the chemical side…
Arrhenius, the founder of physical chemistry, proposed a theory to help differentiate the two.
Acids release H+ ions in their aqueous state, while bases release OH- ions in their aqueous state.
H2SO4 (aq) → 2H+ + SO42- = acid! (diprotic)
NaOH (aq) → Na+ (aq) + OH- (aq) = base!
However, certain substances will show acidic or basic properties regardless of being in an aqueous state or not, hence, his theory isn’t applicable anywhere. For example, CO2 is still acidic despite being a gas. So we now turn to…
The Bronsted-Lowry Theory
Here, acids are “proton donors”, meaning they give out H+ ions, which are basically just protons. A hydrogen atom has one proton and one electron with zero neutrons. Strip away the electron to get an H+ ion, and Bob’s your uncle, you have a proton.
Acids have an excess of H+ ions.
Meanwhile, bases are “proton receivers”, meaning they can accept H+ ions, OR they have an excess of OH- ions. Bases that are soluble in water are called alkali. Remember that not all bases are alkalis (that is to say, not all bases are soluble in water, but all alkalis are bases!)
[insert conjugate.acid.base.png]
Conjugate Acid-Base Pair
Conjugate acid-base pairs are pairs with a single proton difference, or the difference of an H+ ion. Not two, not three, just one.
HF (hydrogen fluoride) is the acid here, because it is a proton donor and gives up a proton, causing it to become negatively charged fluorine (because bye bye hydrogen!). This makes the F- a conjugate base. F- now can accept a proton.
Meanwhile, H2O is the base here because it accepts the proton, causing it to become positively charged H3O (because hello hydrogen), making H3O a conjugate acid. H3O can now give up a proton.
The charges of the ions change because atoms can have their charges changed in two ways: either by an increase or decrease in electrons, or an increase or decrease in the number of protons. Here, it’s with protons!
Note that some chemical compounds can be both conjugate acids and bases in different conditions, like NH3
If we take it as a base:
(here, NH3 is being greedy and acting as a proton acceptor, stealing an H+ ion from water)
If we take it as an acid:
(here, NH3 is being generous and giving up an H+ ion to water)
Universal Indicator
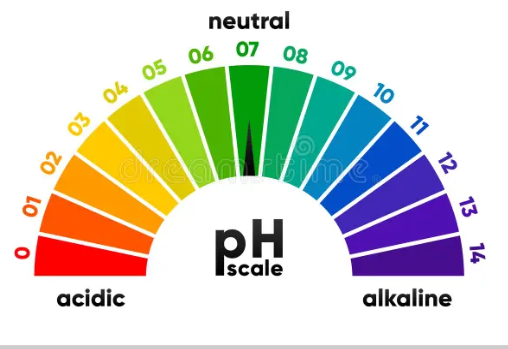
This is a chemical that contains a mixture of multiple indicators which gives a range of colour when added to different acids and bases. It gives a rough idea of how strong an acid or base is. The pH scale colour will shift from red (acidic) to purple (basic), depending on the nature of the substance.
All about Acids!
Acids can be weak or strong.
Strong Acids:
Dissociate completely, more acidic, lower pH, think HCl, H2SO4.
Weak Acids:
Dissociate partially, less acidic, higher pH, think acetic acid.

“Dissociation” refers to them breaking down into ions. Weak acids will mostly stay as molecules and not break down into ions. To figure out whether an acid is weak or strong, we can use several distinguishing tests, such as:
Tests to Distinguish Strong and Weak Acids
- Conductivity Test: Stronger acids are better conductors because they produce more ions due to complete dissociation.
- pH Test: Strong acids have a pH of 2 or less, while weak acids have a pH of 2–7.
- Rate of Reaction: Stronger acids are more highly reactive because they produce more ions and can react fully with bases.
Reactions of Acids
These are super important, so try to memorize them to save time in the exam.
- Acid + Base → Salt + Water
- Acid + Metal Hydroxide → Salt + Water
- Acid + Metal Oxide → Salt + Water
- Acid + Metal Carbonate (CO3) → Salt + Water + Carbon Dioxide (CO2)
- Acid + Metal Hydrogen Carbonate → Salt + Water + CO2
- Acid + Metal → Salt + Hydrogen Gas (produces “effervescence”)
Note: Water is only produced if there is a hydroxide ion, oxide, carbonate, or hydrogen carbonate present.
Example: Sulfuric Acid and Copper(II) Oxide
Break down the reactants into their constituent ions:
CuO → Cu2+ + O2-
React the first cation with the second anion and the first anion with the second cation:
SO42- + Cu2+ → CuSO4
Write down the products and adjust coefficients as necessary:
To test for CO2, you can do the flame test (place a candle in gas; if it goes out, it’s CO2) or the limewater test (CO2 + Ca(OH)2 → CaCO3, turning the limewater milky).
pH Scale
pH stands for “Power of Hydrogen” or “Potential of Hydrogen”. It measures the hydrogen ion concentration per liter of solution:
For bases, pOH is the “Power of the Hydroxide Ion”:
The more diluted an acid, the higher the pH value. The more diluted a base, the lower the pOH value. Remember:
If pH increases by 1 unit, H+ concentration decreases by 10 units and vice-versa. The same applies for OH-.
Antacids
Used to neutralize excessive stomach acid and treat acid indigestion. Examples include:
- Milk of magnesia (Mg(OH)2, magnesium hydroxide)
- Alka-Seltzer (NaHCO3, sodium bicarbonate)
Nature of Oxides
There are 4 main types:
- Metallic oxides: Basic. React with acids to form salt + water. Example: MgO, Na2O. Dissolve in water to form alkalis (pH 11–14).
- Nonmetal oxides: Acidic. Dissolve in water to form acids (e.g., CO2 + H2O → H2CO3, SO2 + H2O → H2SO3). React with bases to form salt + water (e.g., 2NaOH + CO2 → Na2CO3 + 2H2O).
- Neutral oxides: Do not react with acids or bases (e.g., CO, N2O, H2O). Some can be dangerous, like CO reacting with hemoglobin.
- Amphoteric oxides: React with both acids and bases. Insoluble in water. Examples:
Al2O3 + 2NaOH → H2O + 2NaAlO2
Strength Versus Concentration
The strength of an acid or base depends on how much it dissociates in water according to both the Arrhenius theory and the Bronsted-Lowry theory. Strong acids like HCl will completely dissolve in water to form H+ and Cl- ions, while weak acids like CH3COOH will only partially dissociate into CH3COO- ions and H+ ions.
Dilution
Always add acid to water in small volumes with continuous stirring so that water can gradually absorb the heat from the acid. The stirring evenly distributes the energy from the water molecules reacting with the acid. If you add acid all at once then the water will immediately turn into steam, damaging the beaker.
NEVER add water to acid because the reaction produces steam immediately, which can damage the beaker or cause injury. Adding acid to water is safer because the water can absorb more heat.
Dilution is calculated using the formula:
To calculate the volume of water to add, subtract V1 from V2.
Properties of Bases
- Base + Amphoteric Metal → Salt + Hydrogen
- Base + Nonmetal Oxide → Salt + Water
- Base + Ammonium Salts → Salt + Ammonia (NH3) + Water
Metal oxide bases are soluble in water (e.g., KOH). Transition metal oxide bases are insoluble in water (e.g., CuO).
Salts
Salts are ionic compounds made of positive and negative ions. They are formed when H+ from an acid is replaced by a metal ion from a base or metal. Salts can be soluble (aq) or insoluble (s).
Applications include fertilizers, batteries, fungicides, and health products:
- Potassium chloride (fertilizer, part of NPK)
- Magnesium sulphate (fungicide, epsom salt)
- Calcium carbonate (antacid)
Making Salts with Metals
React metal with acid to produce salt and hydrogen gas:
Making Salts with Insoluble Bases
React insoluble base with acid to produce salt and water. Example with copper oxide:
- Heat diluted acid and stir continuously.
- Add base to acid until solution saturates and precipitate forms.
- Filter solution to remove excess base.
- Evaporate solution to leave behind salt (CuSO4).
Making Salts with Soluble Bases (Alkalis) via Neutralization
React alkali with acid using titration to produce salt and water. Example:
Making Insoluble Salts Through Precipitation
Salts can be soluble or insoluble depending on composition.
| Soluble | Insoluble |
|---|---|
| All sodium (NaCl), potassium (KOH), ammonium (NH4NO3) salts | All nitrates (NaNO3) |
| Chlorides (KCl)… | …except AgCl, PbCl2 |
| Sulphates (Cu2SO4)… | …except CaSO4, BaSO4, PbSO4 |
| Sodium (Na2CO3), potassium (K2CO3), ammonium ((NH4)2CO3) carbonates | …otherwise, all carbonates are insoluble |
Example: Precipitation Reaction for Barium Sulphate
- Mix BaCl2 (aq) and MgSO4 (aq).
- Double displacement reaction forms precipitate (BaSO4) and soluble product (MgCl2).
- Filter to remove BaSO4.
- Wash precipitate to remove soluble impurities.
- Dry precipitate in warm oven.
Criterion B: Reaction of Metals With Acids
Example: Zinc with HCl, H2SO4, CH3COOH (undiluted)
- IV: Strength of acid (HCl > H2SO4 > CH3COOH)
- DV: Rate of reaction measured by disappearance of zinc
- CV: Mass of zinc, volume of acid
Procedure
- Gather materials:
- Acetic acid (CH3COOH) 300 cm3
- Hydrochloric acid (HCl) 300 cm3
- Sulfuric acid (H2SO4) 300 cm3
- Magnesium 10 g chunk
- Beakers 300 ml (3)
- Gloves, tweezers, stopwatch
- Wear gloves and use tweezers for safety.
- Add 100 cm3 of each acid to separate beakers.
- Add 10 g magnesium to each beaker and measure time for complete reaction using a stopwatch.
- Repeat for all acids, three trials per acid, calculate average time.
The experiment can be repeated with copper and dilute acids to compare reactions.
Crit B + C: Catalysts and H2O2
Variables
- Independent Variable: Type of catalyst used (MnO2, Fe2O3, CuO, ZnO)
- Dependent Variable: Rate of decomposition of H2O2 OR volume of foam formed over time
- Control Variables:
- Mass of catalyst used
- Volume of H2O2 added
- Concentration of H2O2
Research Question
How does the type of catalyst added to identical volumes of H2O2 affect the rate of decomposition as measured by the volume of foam formed over time?
Hypothesis
If the type of catalyst used is changed, then the rate of decomposition of H2O2 as measured by the volume of foam produced will change, because the oxidation state of the metal in the catalyst differs.
Manipulation of Variables
- IV: Type of catalyst used, manipulated using catalysts with increasing oxidation states (ZnO & CuO → Fe2O3 → MnO2)
- DV: Rate of decomposition of H2O2 measured by foam volume
- CV1: Mass of catalyst kept constant
- CV2: Volume of H2O2 kept constant
Procedure
- Gather materials:
- 20 ml detergent
- Pipette
- 100 ml measuring cylinders (5)
- 6% H2O2 (100 ml)
- Manganese (IV) oxide, Zinc oxide, Copper (II) oxide, Iron (III) oxide (10 g each)
- Gloves
- Safety:
- Wear gloves to avoid skin contact with H2O2
- Keep measuring cylinder away from face to avoid fumes
- Add 10 g of each catalyst to separate measuring cylinders.
- Observe and record the volume of foam produced over time.
Observation Table
Conclusion
Extension
Real-Life Applications of Acids and Bases
Acids
Acid Rain: Oxides of sulfur and nitrogen dissolve in rainwater:
SO2 + H2O → H2SO3
NO2 + H2O → HNO3
Sources: burning fossil fuels containing sulfur, e.g., at power stations.
Effects: damages crops, makes soil acidic, corrodes buildings (e.g., limestone).
Bases
Neutralize acidic soil using CaCO3 (limestone), CaO (quicklime), Ca(OH)2 (slaked lime/limewater). CaCO3 is preferred as it is insoluble and remains in soil to neutralize acid.
Sink cleaning: Ammonia is used as a drain cleaner.
Experimental Chemistry
Acid can be added to a round-bottom flask using:
- Thistle funnel: extends to bottom of flask, allows acid to flow safely
- Dropping funnel: short tube with knob to adjust release speed, prevents bumping
Gas Collection and Testing
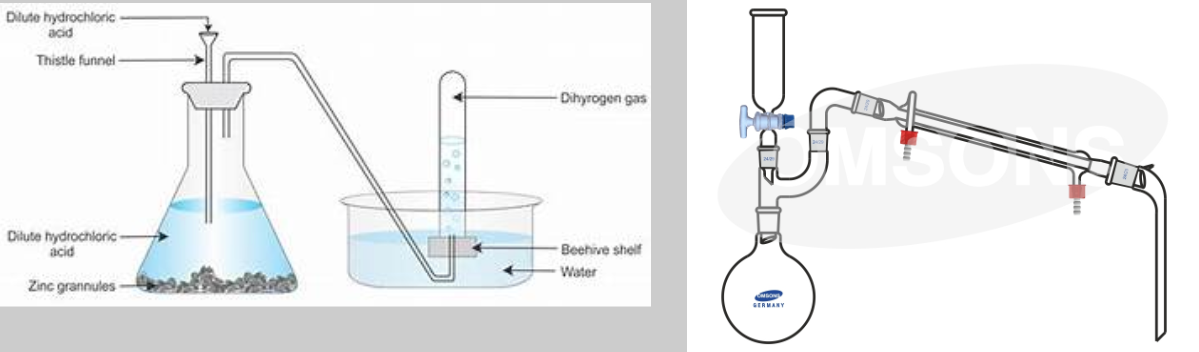
>A thistle funnel versus a dropping funnel setup.
To remove gas produced in a reaction, an exhaust pipe can be inserted into the stopper. There are 4 main methods:
- Upward air displacement (Downward delivery): Collects heavier gases that sink below lighter gases (e.g., HCl, SO2).
- Downward air displacement (Upward delivery): Collects lighter gases that rise above heavier gases (e.g., H2, NH3).
- Over water: For gases insoluble in water (e.g., CO2, O2). Gas passes through water and collects in an upturned jar.
- Gas syringe: Measures the volume of any gas. Gas pressure pushes the plunger to measure volume produced.
Testing for Gases
- Ammonia (NH3): Colourless, pungent, basic. Turns red litmus paper blue as NH3 accepts H+ to form NH4+.
- Carbon dioxide (CO2): Colourless, odorless, mildly acidic. Turns limewater milky (CaCO3 precipitate).
- Chlorine (Cl2): Green, toxic. Bleaches moist pH indicator paper white.
- Hydrogen (H2): Colourless, odorless, explosive with oxygen. Test: lit splint → "pop" sound.
- Oxygen (O2): Colourless, odorless. Glowing splint reignites in pure O2.
Ion Tests
Used to detect cations (+ve ions) or anions (-ve ions) in unknown salts through gas or precipitate formation.
Test for Cations
| Cation | Test | Observation/Signs | Equation |
|---|---|---|---|
| Ammonium (NH4+) | Dilute NaOH + heat | Ammonia gas released, red litmus → blue | NH4+ + OH- → NH3 + H2O |
| Copper (II) (Cu2+) | Dilute NaOH or NH3 | Pale blue ppt; dissolves with excess NH3 → dark blue solution | Cu2+ + 2OH- → Cu(OH)2 [Cu(NH3)4]2+ with excess NH3 |
| Iron (II) (Fe2+) | Dilute NaOH or NH3 | Pale green ppt | Fe2+ + 2OH- → Fe(OH)2 |
| Iron (III) (Fe3+) | Dilute NaOH or NH3 | Red-brown ppt | Fe3+ + 3OH- → Fe(OH)3 |
| Aluminum (Al3+) | Dilute NaOH or NH3 | White ppt; dissolves in excess NaOH → colorless | Al3+ + 3OH- → Al(OH)3 Al(OH)3 + OH- → [Al(OH)4]- |
| Zinc (Zn2+) | Dilute NaOH or NH3 | White ppt; dissolves in excess → colorless | Zn2+ + 2OH- → Zn(OH)2 Excess → [Zn(OH)4]2- or [Zn(NH3)4]2+ |
| Calcium (Ca2+) | Dilute NaOH or NH3 | White ppt with NaOH; little/no ppt with NH3 | Ca2+ + 2OH- → Ca(OH)2 |
Test for Anions
- Halides: Add dilute HNO3 + AgNO3. Chloride → white ppt, Bromide → cream ppt, Iodide → yellow ppt.
- Sulfates: Add dilute HCl + Ba(NO3)2. White ppt of BaSO4 indicates sulfate.
- Nitrates: Add NaOH + Al foil + heat. Ammonia gas released confirms nitrate presence.
- Carbonates: Add dilute HCl; bubble gas through limewater. Milky solution confirms CO2 and carbonate presence.
Types of Water
Water hardness measures dissolved minerals, mainly calcium and magnesium ions, in mg/L or ppm.
- Hard water: Contains calcium + magnesium salts (bicarbonates, chlorides, sulphates). Forms scum with soap, causes scale in pipes and kettles.
- Temporary hardness: Dissolved calcium/magnesium hydrogencarbonates. Removed by boiling.
- Permanent hardness: Dissolved calcium/magnesium sulphates. Cannot be removed by boiling.
Why water becomes hard: Flowing through chalk or lime deposits, water picks up calcium ions.
Organic Chemistry
Topics include:
- Hydrocarbons – saturated and unsaturated, homologous series, characteristics
- Alkanes & Alkenes – nomenclature, preparation, properties, reactions (combustion, substitution, addition, cracking)
- Alcohols & Carboxylic acids – preparation, properties, reactions (esterification)
- Polymers – natural (fats, oils, proteins, carbs) and synthetic (terylene, nylon)
- Soaps and detergents – saponification, hydrogenation of oils, issues with hard water
- New materials – molecular gastronomy, smart materials, nanoworld
- Environmental issues – greenhouse effect, global warming, smog, ozone hole, VOCs, paints & plastic pollution, leaded petrol
Carbon
Carbon is in group 4 of the periodic table and forms up to 4 bonds. Other groups:
- Group 1: 1 bond
- Group 2: 2 bonds
- Group 3: 3 bonds
- Group 5: 3 bonds
- Group 6: 2 bonds
- Group 7: 1 bond
Drawing Lewis Structures
Each line represents a shared pair of electrons. Every element wants 8 electrons in its outer shell (except H: 2 electrons).
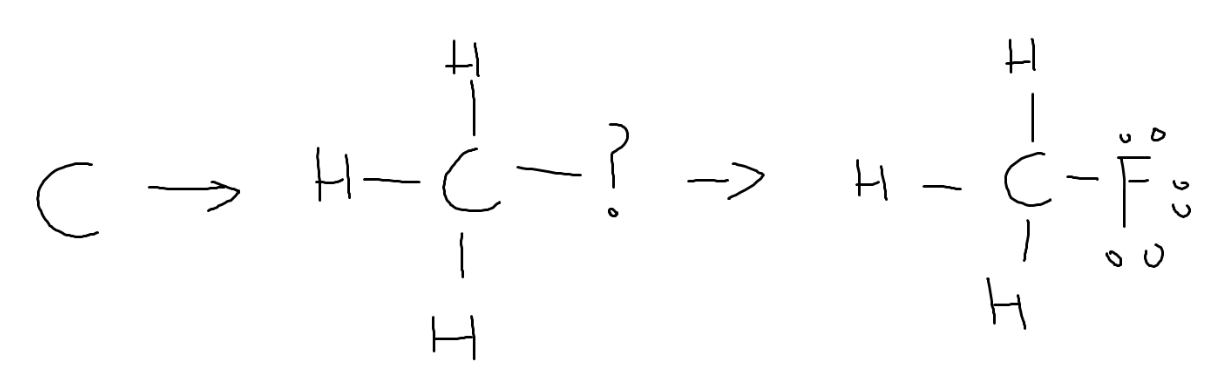
Example: Fluoromethane (CH3F). Carbon forms 4 bonds, each hydrogen 1, fluorine 1. Lone pairs shown on fluorine only.
Alkanes, Alkenes, and Alkynes
| No. of Carbons | Prefix |
|---|---|
| 1 | Meth- |
| 2 | Eth- |
| 3 | Prop- |
| 4 | But- |
| 5 | Pent- |
| 6 | Hex- |
| 7 | Hept- |
| 8 | Oct- |
| 9 | Non- |
| 10 | Dec- |
Alkanes
Saturated hydrocarbons (only single bonds). Formula: CnH2n+2. Example: 4 carbons → C4H10 = butane.
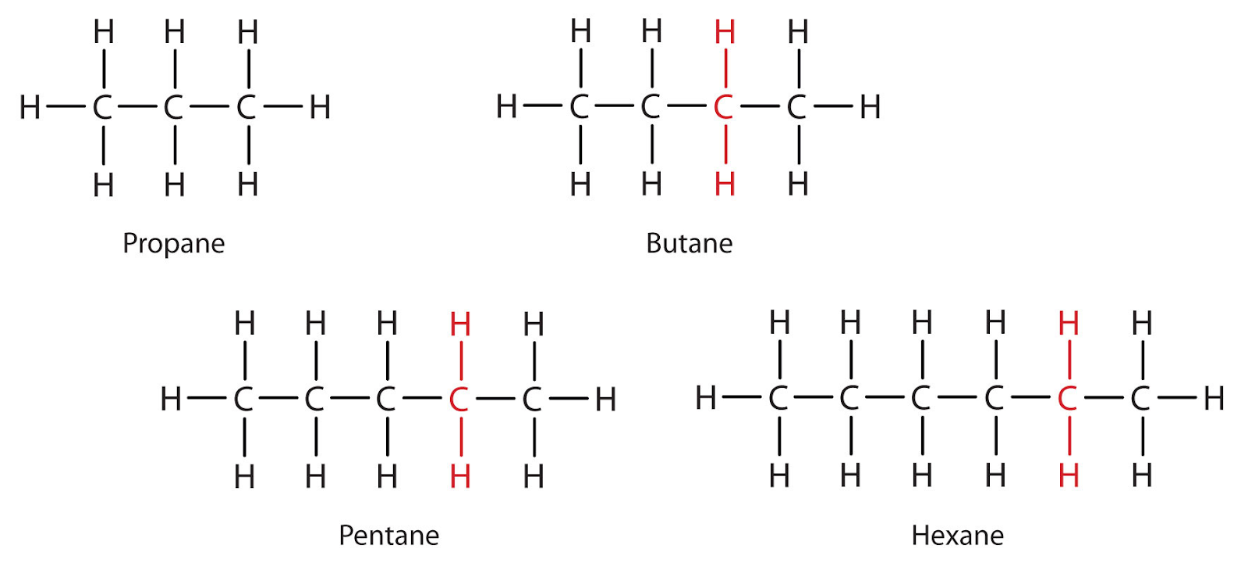
Physical properties:
- Insoluble in water (nonpolar)
- Soluble in nonpolar substances
- Floats on water (density <1 g/mL)
Chemical properties:
- Nonpolar
- Complete combustion → CO2 + H2O + heat
- Incomplete combustion → CO + soot
Alkenes & Alkynes
Unsaturated hydrocarbons with double/triple bonds. Alkenes formula: CnH2n. Example: 4 carbons → C4H8 = butene.
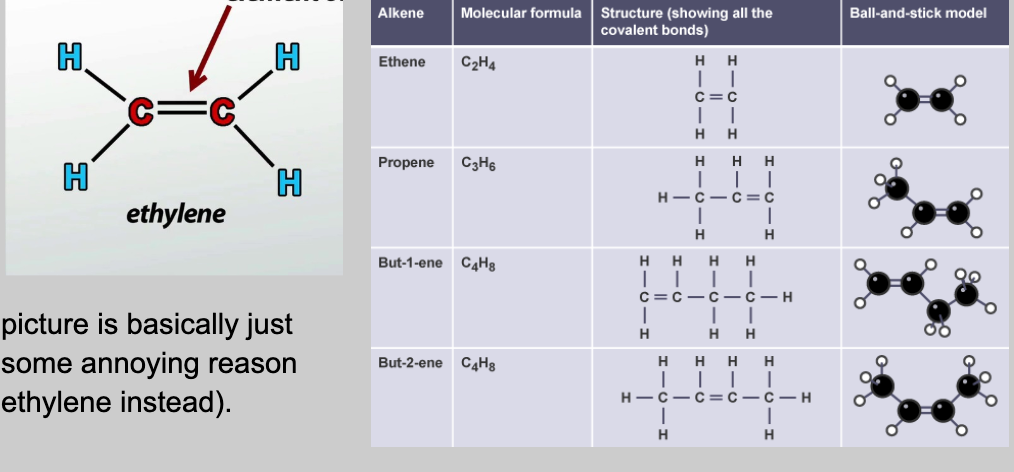
The formula only works if the alkene has a single double bond.
Alkenes Physical Properties
- Insoluble in water: Nonpolar, cannot dissolve in polar solvents like water.
- Soluble in nonpolar compounds: Dissolves nonpolar substances like oil.
- Floats on water: Density < water, explains oil spills.
Alkenes Chemical Properties
- Nonpolar: Mostly covalent bonds, low polarity between C and H.
Alkynes
General formula: CnH2n-2. Example: 4 carbons → C4H6 = butyne.
Rules apply only if there is exactly 1 triple bond. More triple bonds require different naming rules.
Naming Side Chains
Video reference: Click here
Step 1: Find the longest continuous carbon chain.
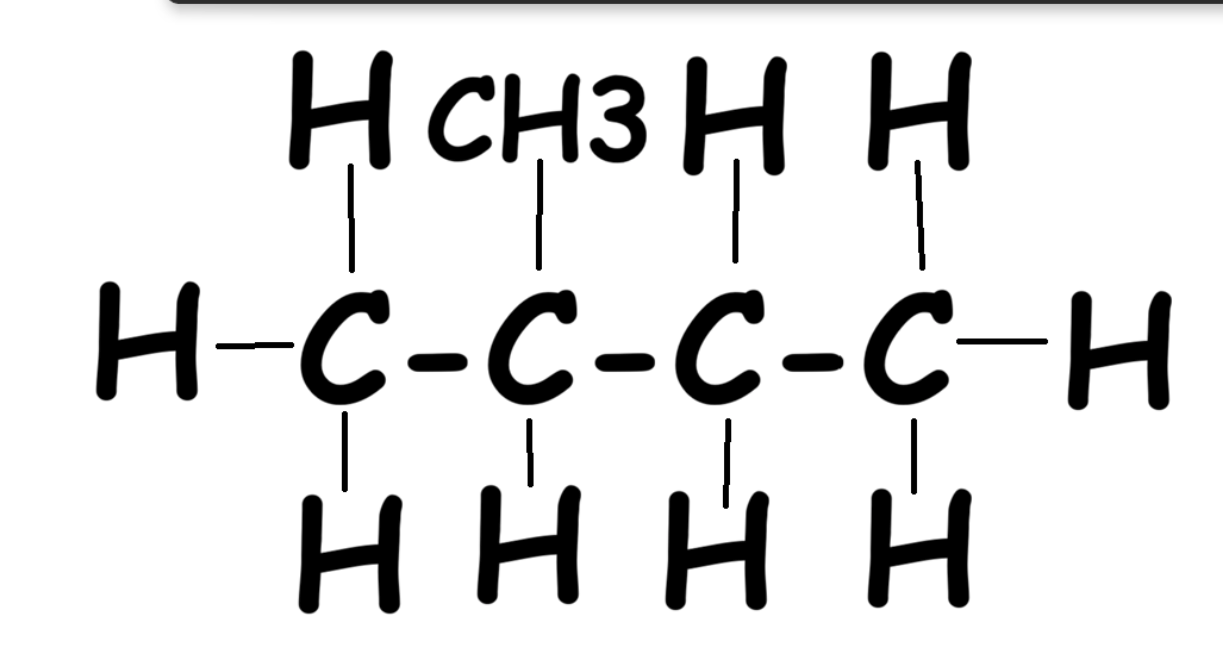
 |
Option #1: 3 carbons long |
 |
Option #2: 4 carbons long |
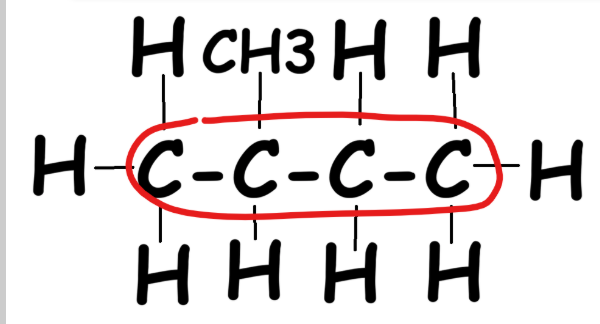 |
Option #3: Also 4 carbons long |
 |
Cat: 3 carbons long |
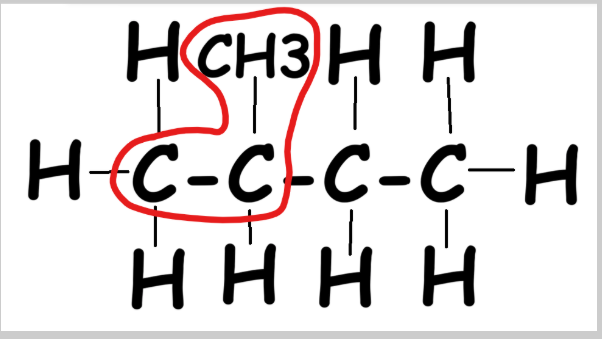
The two longest chains are both 4 carbons → name = butane (single bonds).
Step 2: Number the chain forwards and backwards. Pick the least-numbered chain. CH3 is attached to the 2nd carbon → methyl group.
Compound name: 2-methyl butane
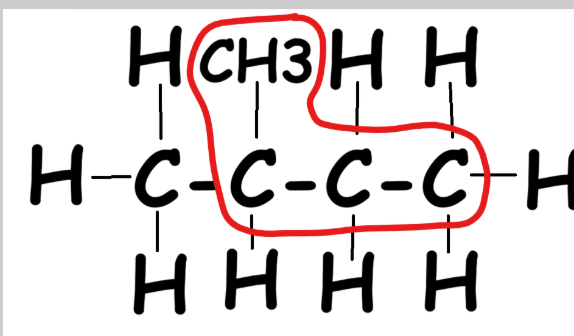
Multiple Side Chains
Follow same rules: find the longest chain, number forwards/backwards, pick least numbers. Example with two CH3 groups:
- Left to right: CH3 at carbons 2 and 3
- Right to left: CH3 also at 2 and 3
- Use smaller number first → 2,3-
- Two alkyl groups → prefix "di" → dimethyl
Final name: 2,3-dimethyl butane
Adding Halogens
Example compound:
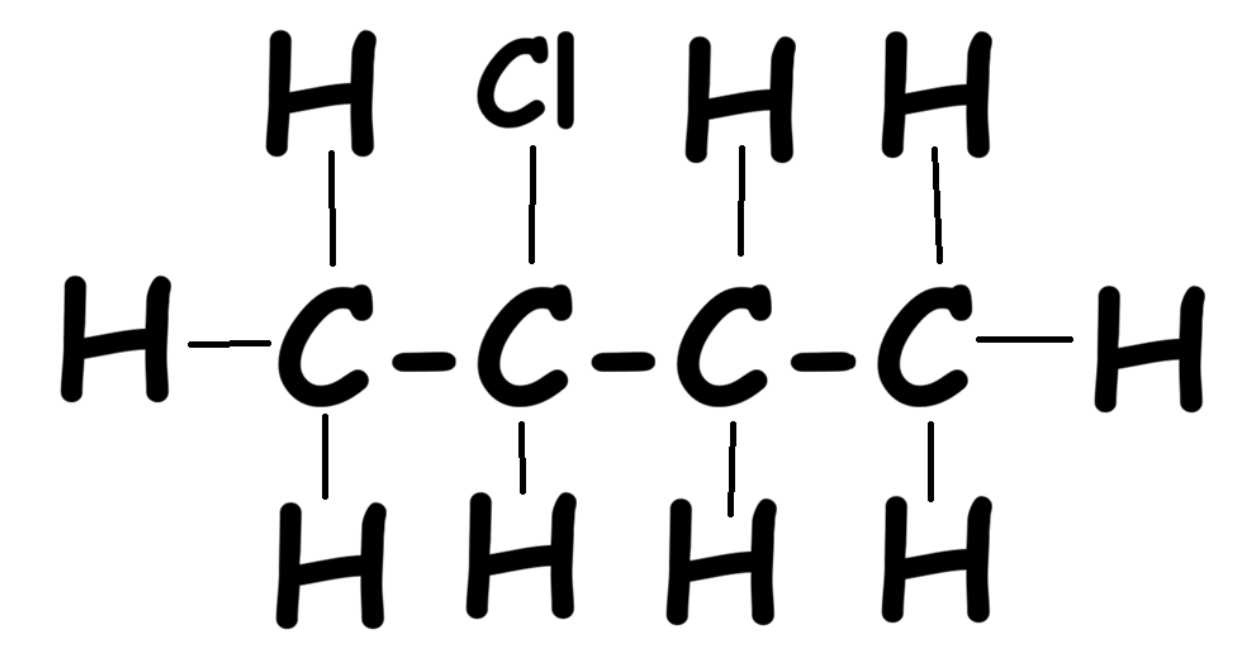
Halogens in Organic Compounds
Instead of alkyl groups, halogens like chlorine can be attached. Use prefixes:
- Chlorine = chloro–
- Bromine = bromo–
- Fluorine = fluoro–
- Iodine = iodo–
Steps to name:
- Find the longest carbon chain.
- Number the chain forwards and backwards.
- Pick the least-numbered chain.
- Name the compound using halogen prefix + chain name.
Example with one chlorine: 2-chloro butane
Example with multiple halogens: alphabetical order → 1-bromo 2,3-dichloro butane
Naming Alkenes
Locate double bond, number to give lowest position. Example: double bond between carbons 2 and 3 → but-2-ene or 2-butene
Naming Alkynes
Locate triple bonds, number for lowest position. Two triple bonds → suffix –diyne. Example: triple bonds at carbons 1 and 3 → pent-1,3-diyne
Alcohols and Alkyls
Alkyls are groups with one less hydrogen than alkanes. Suffix: –yl
| Alkane | Formula | Alkyl |
|---|---|---|
| Methane | CH4 | CH3 (methyl) |
| Ethane | C2H6 | C2H5 (ethyl) |
| Propane | C3H8 | C3H7 (propyl) |
Homologous Series
Members share the same functional group and differ by CH2 units.
- Alkanes, alkenes, alkynes, alcohols, carboxylic acids etc.
- Same chemical properties (functional group), different physical properties (mass, boiling/melting point).
| Alkane | Alkene | Alkyne |
|---|---|---|
| CH4 (methane) | CH2 (methylene) | C2H2 (ethyne) |
| C2H6 (ethane) | C2H4 (ethylene) | C3H4 (propyne) |
| Difference: CH2 | Difference: CH2 | Difference: CH2 |
Fractional Distillation of Crude Oil
Video reference: Click here
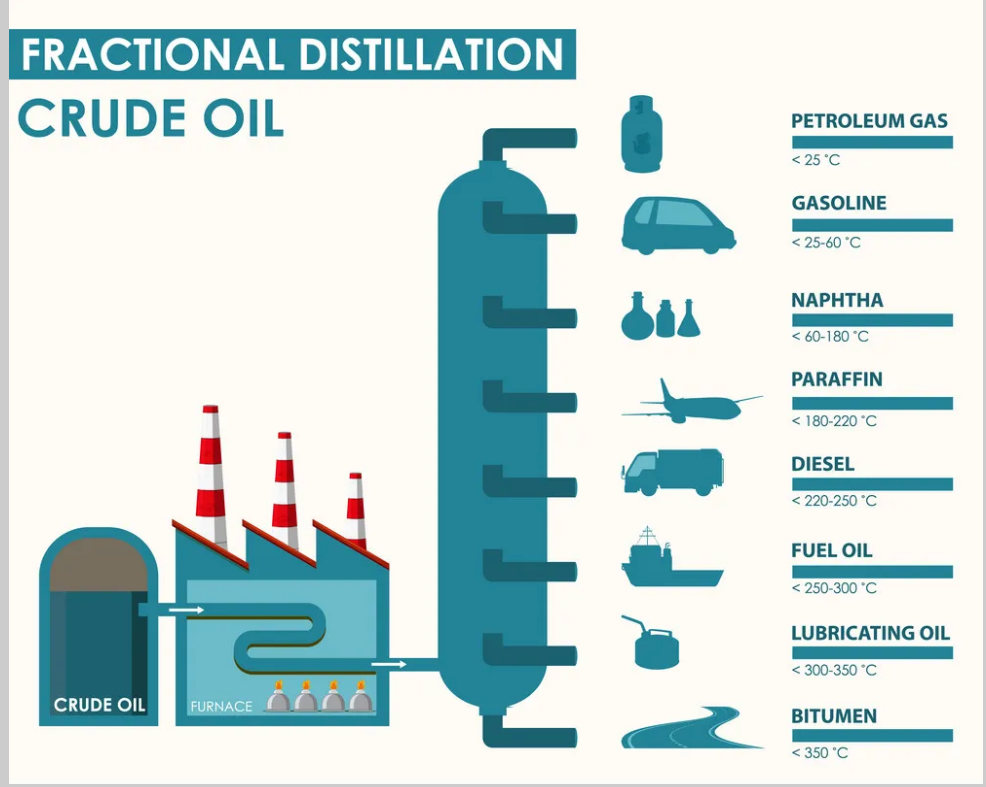
Crude Oil
Crude oil is unprocessed oil, a mixture of hydrocarbons with varying chain lengths. Long-chain hydrocarbons have stronger London Dispersion forces and higher boiling points, making them harder to separate. Short-chain hydrocarbons have lower boiling points and are more valuable as fuels.
Fractional Distillation
Crude oil is heated into vapor and pumped into a fractionating column with a heat gradient (hot bottom, cooler top). Long-chain hydrocarbons condense lower in the column, while short-chain hydrocarbons condense higher. Bubble caps prevent mixing and encourage condensation. Each tray contains fractions with similar boiling points, used for different purposes like fuels or bitumen for roads.
Cracking
Long-chain hydrocarbons can be broken into smaller alkanes and alkenes. No fixed formula exists, but the number of atoms in the products equals the reactant.
- Thermal cracking: 750°C, 70 atm
- Catalytic cracking: 500°C, zeolite catalyst
Functional Groups
Functional groups define chemical properties of hydrocarbons:
- Alcohols
- Carboxylic acids
- Amides: Dicarboxylic Acid + Diamine → CONH linkage
- Esters: Dicarboxylic Acid + ...
Alcohols

Alcohols are organic compounds with an –OH group attached to them, which is their functional group. Like alkanes, alkenes, and alkynes, they form a homologous series, too! (Methanol, ethanol, propanol, butanol, etc.)
When naming alcohols with just an OH group, we replace the end “e” with an “ol” (Methane → MethanOL, etc.) For naming more complex alcohols, refer to previous topics.
The general formula for these guys is CnH2n+1OH.
this will be below the image
CH(2+1)OH
=CH3OH
…Which gives us methanol!
Remember, the –OH always goes at the end of each formula.
Making Alcohols:
Fermentation: The good, old-fashioned, and environmentally-friendly way to make alcohol. This process involves breaking down glucose without the presence of oxygen, which gives rise to alcohol, CO2, and ATP, as you would’ve studied in biology.
For this, natural sugar sources are used, such as grapes for red wine, and barley for beer. This process takes an extremely long time, but can be sped up with the catalyst of yeast, a natural fungus that feeds on sugar and produces carbon dioxide. This is also why bread has holes– they come from the carbon dioxide bubbles that burst during baking!.
It can also be done at temperatures as low as 37 degrees Celsius, and since plant sugars are a renewable resource, it’s good for the environment! However, it forms the waste product of carbon dioxide.
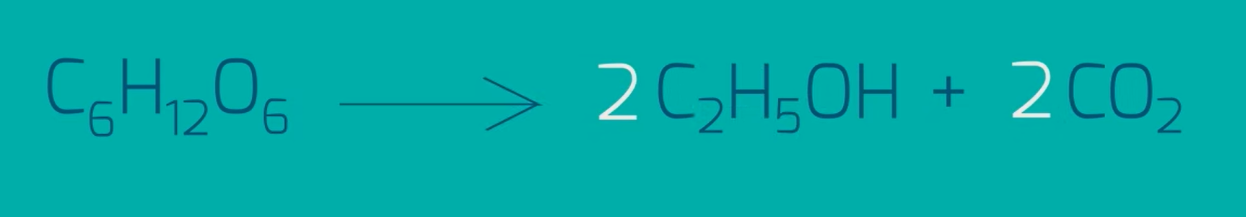
Hydration of Ethene: This one’s a different route with a similar ending. Ethene is an alkene extracted from the cracking of crude oil. Crude oil is a non-renewable resource, so this way isn’t exactly sustainable.
Ethene undergoes an addition reaction with water as steam (hydration) and phosphoric acid as a catalyst, forming just ethanol with no waste products! However, it has to be heated to extremely high temperatures to do this, making it a costly and difficult process.
Physical Properties
High Boiling Point: Alcohols have a high boiling point due to the hydrogen bonds formed between molecules thanks to the –OH group.
Chemical Properties

Flammable: You’ve likely heard of a Molotov cocktail, a weapon where a glass bottle filled with alcohol and a fuse, has the fuse lit and then thrown. The bottle shatters on impact, releasing (literally) flaming-hot alcohol and glass shards. This is all thanks to the fact that alcohol is flammable!
Alcohol reacts with oxygen to produce carbon dioxide and water, undergoing complete combustion.
Soluble in Water: Alcohol molecules are polar, meaning they dissolve in polar solvents like water.
Form Neutral Solutions: Alcohols are neither alkaline nor acidic, meaning the solutions they form upon dissolving in water have a neutral pH.
Form Carboxylic Acids: Upon adding an extra oxygen to an alcohol (oxidation), we get the functional group –COOH, which belongs to carboxylic acids!
Uses of Alcohols
Weapons: Two words. Molotov. Cocktail.
Fuels: Due to its flammable nature, alcohol can be combusted and used as a fuel that releases a lot of heat energy. Examples include spirit burners, which are used in place of Bunsen burners at times. While the latter runs on natural gas, the former uses alcohol as a fuel to burn.
Drinking: This is self-explanatory. Yo ho ho and a bottle of rum!
Industrial Solvents: Alcohol is often used because it dissolves things that water can’t, like lipids such as oil, as well as hydrocarbons.
Antiseptics: Don’t drink your sanitizer, kids! “Rubbing alcohol”, or alcohol at extremely high concentrations, is often used for sterilization of wounds and cuts. This is for a variety of reasons. Firstly, alcohols dissolve lipids, meaning they could disrupt the cell membrane of microorganisms. Secondly, they damage proteins in germs by breaking apart the bonds and forming hydrogen bonds with the molecules in the protein, denaturing them. They also evaporate quickly, meaning you don’t have to wipe your hands, risking exposure to more germs.
Esters
Esters are versatile organic compounds characterised by their functional group –COO, with a carbon double-bonded to one oxygen and single-bonded to the other. They are found in perfumes, natural fragrances, plastics, and even lipids and fats in the body!
They have an excellent fragrance, hence their usage in perfumes.
They are also volatile, which sounds dangerous, but really just means they evaporate quickly and easily, making them good for perfumes.
Esterification of Alcohols/Fischer Esterification:
In order to form esters, a process known as Fischer esterification is undertaken. In this process, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, usually sulfuric acid, to produce an ester and water. The carboxylic acid gives up one hydrogen, which reacts with the alcohol to produce water. Meanwhile, the rest of the molecule attached to the alcohol, denoted by an “R”, bonds with the carboxylic acid, forming an ester.
This reaction is therefore a condensation reaction, as two molecules join together to form one molecule, and a small water molecule is released.
The reaction, however, is reversible! Reacting an ester with water, in exchange, gives you a carboxylic acid and an alcohol.
Uses:
- Perfumes
- Food flavorings
Carboxylic Acids
These are organic compounds with a functional group of –COOH, which form a homologous series (methanoic acid, ethanoic acid, propanoic acid, and butanoic acid). Furthermore, all the acids’ names end in “--anoic acid”, followed by the prefix depending on the number of carbons (1 carbon = methanoic acid).
!!IMPORTANT!! Note that the “C” in –COOH is counted among the carbons, despite being part of the functional group.
When writing their formulas, the –COOH goes at the end.
Making Carboxylic Acids:
Carboxylic acids are formed by the oxidation of alcohols. Alcohols already have the functional group –OH, and likely have a carbon atom somewhere. Thus, adding extra oxygen produces –COOH!
The carboxylic acid, if it has the same number of carbons as the alcohol it was made from, will adopt the same prefix as it. For example, butanoic acid made from butanol and oxygen has the same prefix.
Carboxylic acids have a high boiling point due to strong hydrogen bonds between molecules. As their chain length increases, though, their solubility in water decreases.
Physical Properties:
Colorless.
Foul-smelling: Ever wonder why locker rooms and gyms smell so bad? That’s because sebum, a compound naturally produced by the skin, contains carboxylic acids, which have notoriously bad odors.
Chemical Properties:
Weak Acids: Though this is in organic chemistry, carboxylic acids are still acids, a.k.a hydrogen donors! However, they are very weak acids, meaning they don’t fully ionize and give up their hydrogen ions, hence; the process is depicted as reversible.
The negative ion is named such that it ends in “--anoate”, with the prefix depending on the number of carbons in the initial acid.
Reactions with Metal Carbonates: Acids react with metal carbonates to produce water, salt, and carbon dioxide.
Reactions with Bases: Carboxylic acids react with bases to produce a salt and water.
Reaction with Metals: Carboxylic acids react with metals to produce salt and hydrogen.
Natural Polymers
If you need a little pick-me-up on polymers, check out the section in Grade 9 Biology. Some common and crucial biomolecules include lipids, proteins, and carbohydrates.
Proteins:
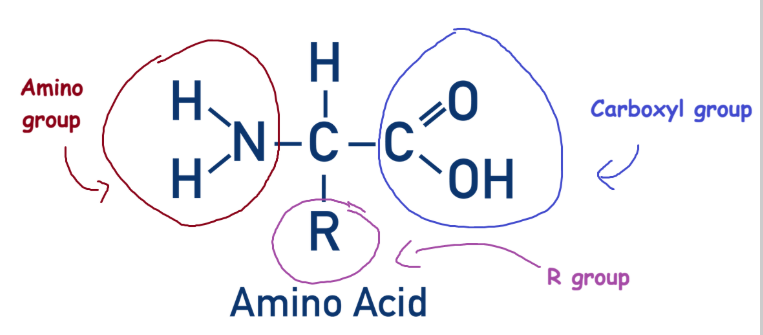
A protein is a polymer formed by multiple folded polypeptides, which are chains of amino acids. Amino acids themselves are made of three main groups: the amino group, the carboxyl/carboxylic acid group, and the R group.
The amino group and carboxyl groups are joined by a central carbon, and are found across all amino acids. The R group, however, changes depending on which amino acid it is.
Amino acids are joined together by amide/peptide bonds, which are formed through a condensation reaction.
Here, the amino group on one amino acid gives up one hydrogen, while the carboxyl group on the other amino acid gives up one hydrogen and one oxygen. This forms one molecule of water, while the remaining nitrogen and carbon bond together.
Carbs:
Carbohydrates are polysaccharides, meaning they are polymers formed from multiple monosaccharides, or subunits called glucose. They contain only hydrogen, oxygen, and carbon, hence the name CARBO-HYDRATES (carbo = carbon, hydrates = hydrogen and oxygen).
The oxygen atom in one unit of glucose becomes extremely electronegative and becomes attracted to the carbon atom in the other unit of glucose, that has a partially positive charge. Thus, the oxygen breaks its bonds with the –OH group attached to it, and bonds with the carbon, creating a disaccharide that can later become a polysaccharide. Meanwhile, the –OH group bonds with an H+ ion to form water!
Lipids:
Now, lipids are a diverse group and don’t necessarily have a single, consistent monomer, but a very common type of lipid, a triglyceride, consists of glycerol, an alcohol with –OH groups (triol) and fatty acids (carboxylic acids that are either saturated or unsaturated).
Saturated fatty acids consist of carboxylic acids joined entirely by single bonds, meaning every single carbon is bonded to a hydrogen, similar to alkanes! Saturated fats are solid at room temperature, such as butter and cheese. The straight-chain structure of these bad boys means they can easily pack together tightly, hence their solid state at room temp.
Unsaturated fatty acids consist of carboxylic acids that contain at least one double bond between carbons, meaning not all carbons are bonded to hydrogens. Those with just a single double bond are called monounsaturated fatty acids, while those with more than one are polyunsaturated fatty acids. These are found in nuts, avocados, and olive oil, and are liquid at room temperature. The presence of these double bonds causes “kinks” or bends in the structure of the fatty acids, and they lower LDL cholesterol and increase HDL cholesterol. These very kinks and bends prevent them from packing together as tightly as saturated fatty acids, hence, they are liquid.
In general, saturated fatty acids are more unhealthy than unsaturated fatty acids because they raise LDL cholesterol levels and are linked to heart disease, but a person’s overall intake of fat should be limited to live a healthy lifestyle.
Synthetic Polymers
Terylene is a type of polyester, which means at least one ester linkage is found between repeating monomers. On that basis, terylene is similar to lipids as they both have ester linkages, except that the former is a synthetic polymer, and the latter is a natural one.
Terylene is produced from a dicarboxylic acid (a carboxylic acid unit with two –COOH groups) and a diol (an alcohol unit with two –OH groups).
Properties:
Wrinkle- and shrink-resistant: Some natural fabrics like wool may shrink after multiple washes, but terylene doesn’t. It also doesn’t have wrinkles, meaning it can be blended with natural fabrics for the best of both worlds.
Rigid and durable: Terylene is incredibly durable and rigid, meaning it has many applications in industry and adventure sports.
Hydrophobic: Terylene repels water rather than accepts it, making it resistant to liquid stains as well as keeping the wearer dry.
High melting point: Terylene only melts at extremely high temperatures, making it a solid choice for industrial applications.
However, it’s costly to manufacture and bad for the environment as it isn’t biodegradable.
Uses:
- Raincoats (because it’s hydrophobic)
- Sails and nets (due to hydrophobic properties and durability)
- Plastic bottles and packaging (it’s a polyester, just like PET)
Nylon was developed by DuPont de Nemours, an American chemical company, in 1935, particularly by a man named Wallace Carothers. Despite being a wildly successful product today, Carothers took his own life just 16 months before its release onto the market due to chronic depression, never being able to see its popularity.
Nylon belongs to the family of polyamides, similar to proteins, as subunits are joined together by amide linkages.
Both of these polymers are formed through condensation polymerization, which is one of two ways synthetic polymers are formed. The other way synthetic polymers are formed is through addition reactions, like polyethylene (polyethene), polypropylene, and PVC (polyvinylchloride).
Because they are joined by amide linkages, nylon is similar to proteins! The only difference? Proteins are natural polymers, and nylon is synthetic.
Properties:
Strong (Nylon is one of the strongest fibres, making it incredibly durable and thus it can be used in industrial settings).
Lightweight (This, in combination with its strength, is another factor behind its use in industry).
Hydrophobic.
Resistant to alkalis (Though repeated exposure in large concentrations will eventually damage the fabric, nylon is more resistant to alkalis than acids).
However, white nylon can become yellow over time due to sunlight exposure, damaging the color. Furthermore, the fabric isn’t very breathable.
Uses:
- Clothing: Nylon is often mixed with other natural fabrics or even artificial ones like polyester
- Rope: Nylon is strong despite its light weight, making it ideal for climbing ropes
- Parachutes: Lightweight so it doesn’t weigh the person down, but strong enough to hold weight
- Tents/Tarpaulin Sheets: Waterproof and lightweight for outdoor use
Saponification, a.k.a Hydrolysis!
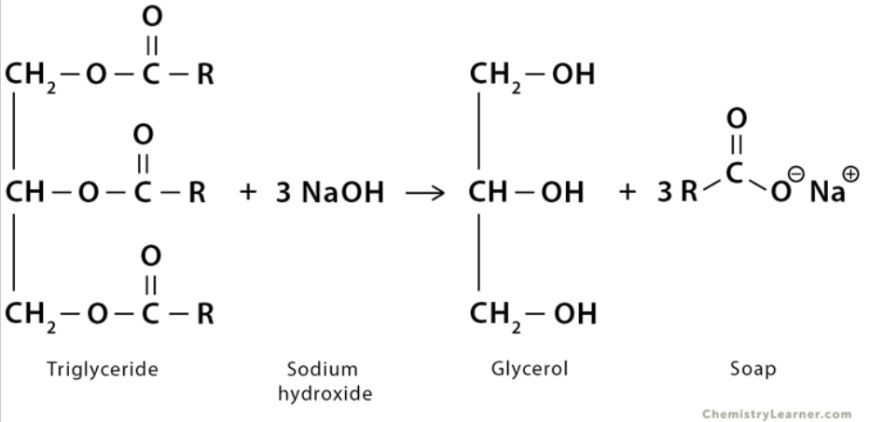
The term “saponification” comes from the Latin word “sapo”, which means soap. It’s an accurate moniker that essentially translates to the creation of soap, which is what happens in this process.
Saponification is the process of breaking down esters using water and an aqueous alkali into an alcohol and a salt of the metal and the carboxylic acid.
The alkali used are typically ones like potassium hydroxide or sodium hydroxide, that are soluble in water. Meanwhile, the most common esters used are triglycerides (triesters). Since ancient times, triglycerides have been found in animal fat. It’s believed the ancient Babylonians would boil animal fat with ashes. The ashes themselves contained potassium carbonate, which was an alkali. This would break down the fat into soap, which was then wetted and used to wash pots and pans.
The alcohol that is usually produced is called glycerol, while the salt becomes the soap. Glycerol is further purified to create glycerin, which is often added to make soaps translucent.
Oftentimes, alcohols such as ethanol are added to help the oil dissolve better in the water, as the two compounds often don’t mix.
Oil and Water With Ethanol vs Without:

In a sense, this reaction is the opposite of a condensation reaction, because here, a molecule is being split using water.
The rate of the reaction can be increased by increasing the temperature by placing it in a hot water bath. Take safety precautions when doing this with concentrated sodium hydroxide, as in a concentrated form, it can damage the eyes and skin, so wear gloves and goggles!
Finally, we can precipitate the soap from the glycerol by “salting it out”.
Salting Out
In this process, we decrease the solubility of the solvent the soap is in by adding a large volume of salt, and therefore, the soap becomes less soluble and rises to form a precipitate.
To put it simply, normally, when soap is soluble, the compounds in the soap have a certain charge. Water molecules form hydrogen bonds accordingly, and surround the molecules, dissolving it. However, if we add a large volume of NaCl (sodium chloride), the solvent becomes more attracted to it, dissolving the NaCl instead of the proteins. Thus, the proteins form a precipitate.
This precipitate can then be filtered out using a typical apparatus of a funnel and filter paper, where the glycerol is the filtrate and the soap is the residue.
To test whether what you’ve made is, indeed, soap, and not some kind of biological weapon, simply take a spatula-ful of soap, add it to a test tube with water, cork it, and shake it. If it forms a lather, congratulations! You have soap.
Soap and Detergent
Soap and Detergent
Soaps are the salts of fatty acids produced through saponification used for cleaning purposes or lubrication. There are two types of soap:
Hard soaps are solid at room temperature, and are formed from using sodium hydroxide as the alkali during saponification. These are bar soaps and round soaps. They dissolve slowly. This is because the lipid it is produced from contains esters with extremely long hydrocarbon chains attached to the ester functional groups, typically with 12 or more carbons, which are harder to break apart when heated, meaning they are solid at room temperature.
Soft soaps are liquid at room temperature, and are formed from using potassium hydroxide as the alkali during saponification. These are often the soaps that come in bottles. They dissolve quickly.
However, one thing you’ll notice across soap brands is that they never use esters with six or less carbons in a hydrocarbon chain, as they irritate the skin and smell bad.
How Does Soap Work?
Most compounds are usually either hydrophobic or hydrophilic, meaning they are either attracted to water or are repelled by it. There are, though, certain compounds that contain both hydrophobic AND hydrophilic, including soap and detergent. Soap, especially, is a surfactant, meaning it lowers the surface tension between compounds in two different phases, like between solids and liquids, or liquids and gases.
What is Surface Tension?
Surface tension is the tendency of liquids to shrink into the form with minimum surface area at rest. This is why water forms circular droplets, as they have the least surface area. This is caused by the uneven forces acting on water molecules. Molecules deep in the water have force acting on them from all directions, meaning there is no net force on them. However, molecules on the surface of the water don’t have any force acting on them from above, meaning they experience a stronger net down force, as well as a cohesive force between molecules next to it, causing to be “pulled in”, increasing the density of water droplets at the surface and pulling the water into a spherical shape.
But how is this relevant to the functioning of soap, you may ask?
Well, soap can’t wash dirt, oil, and other stains away just like that. It needs to form an emulsion with the substance and water, and with things like oil, it’s practically impossible. It’s even harder when the water keeps beading up, because then it won’t be able to fully absorb and wash away the stains with the soap molecules. Surfactants help by getting in the way! See, soap molecules contain a hydrophobic, nonpolar “tail” and a hydrophilic, polar “head”. This means that if a soap molecule enters water, the hydrophilic head will readily dissolve in water, but the hydrophobic tail nopes right out of there, preferring to stick out in the air.
This causes the molecule to position itself on the water’s surface for the optimal, lowest-energy state. As a result, though, the molecule wedges itself between the water molecules, reducing the cohesive energy between them and causing the water to spread out! This reduces surface tension!
How Do Surfactants Reduce Surface Tension?
The main action of soap depends on its ability to form micelles, which are spherical structures formed when surfactant molecules accumulate in one place.
Let’s say we’re trying to wash a shirt with oil stains on it. The oil will obviously be insoluble in water, and will just remain in the shirt. Time to bring out the soap! Now, the hydrophilic soap molecules obviously are attracted to the water, but the hydrophobic soap molecules are nonpolar, and so they dissolve in oil instead! This forms a spherical structure with the heads facing “out” into the water and the tails facing “in” to the oil.
Now, this happens at such a small scale that it appears the oil and water have mixed together, when in reality, a simple colloid has been formed. Now, when the water heads down the drain, it pulls the hydrophilic heads with it. These heads pull the tails with them, and the tails pull the oil molecules. And voila, your shirt is clean! And wet. You should probably hang it out to dry.
A similar thing happens within your cell membrane! That’s why it’s called a bilayer, semi-permeable membrane, because it’s a bilayer of lipids, with the heads dissolving in the water cytoplasm as well as the area outside the cell, and the tails in between being sandwiched.
Soaps v.s. Detergents
Detergents are similar to soaps and perform the same function. However, soaps are synthesized from natural ingredients like animal fat or vegetable oil, while detergents are made from synthetic chemicals and salts of stronger acids than of soaps.
Soaps also dissolve slower and easily biodegrade, making them better for the environment but slow at their job. They are also gentler, hence why they’re used by humans when bathing. Detergents, on the other hand, only have a few options that biodegrade, but dissolve much faster and are much harsher, which is why you want it to clean your clothes, but not what lies under your clothes. Don’t bathe with detergent, kids!
Soap and Hard Water
How Does Hard Water Affect Soap? - Biology Insights
If you don’t remember, go have a refresher on what hard water is. In short, hard water is water with a large concentration of dissolved mineral ions. Mainly groundwater, containing Ca2+ and Mg2+.
Now, typically, the hydrophilic head of the soap, that is negatively charged, has a positively-charged ion such as Na+ or K+ with it that enables it to dissolve in water, as the positive ions are attracted to the partially negative oxygen atoms.
However, Ca2+ and Mg2+ are more attracted to the negative head of the soap than the original Na+ or K+, which are displaced. As a result, two soap molecules bond to each ion, forming compounds insoluble in water like calcium/magnesium stearate. Thus, the stains are neither cleaned, as the soap never dissolves in water in the first place. The stearate precipitate forms a whitish-grey scum.
This has two consequences:
Things stay dirty: Like I said, the soap is too busy forming insoluble compounds to react with stains and form micelles, meaning the thing you want to wash never gets clean.
Furthermore, the only way to make the soap work with hard water is to keep adding soap till all the ions have been reacted with, so that it can finally form micelles.
Things get dirtier: This scum formed is sticky, sticking to showers, drains, walls, sinks, and even clothes! This creates a filthy-looking residue that’s very hard to get out.
However, we can combat this problem by using strong, synthetic detergent. Due to its different composition, detergents remain water soluble despite bonding to the ions in hard water, meaning things can still be cleaned as micelles are still formed, as long as they are in the water, they can still bind with dirt molecules and form micelles, compared to the insoluble soap scum that just forms a useless precipitate.
Hydrogenation of Oils
Remember those lipids we talked about earlier? Yeah, well, like I said, there’s saturated (alkanes) and unsaturated (alkenes) fats. Wanna know how to make an unsaturated one into a saturated one?
It’s simple– the process is called hydrogenation. It involves adding, you guessed it, hydrogen gas to unsaturated fats.
It’s not as easy as that, though. You have to heat the unsaturated fat to 60 degrees Celsius alongside a platinum catalyst before passing hydrogen gas through it.
Now, with polyunsaturated oils, you don’t have to hydrogenate all the bonds. The more you hydrogenate, the harder in texture it becomes and the more solid it becomes, the less you hydrogenate, the more liquid and softer it remains.
Materials (Nelson Textbook Topic #4– Materials!)
Kevlar
Kevlar: A polyamide stronger than steel, used to make bulletproof vests and brake pads in cars.
Spandex (Lycra / Elastane)
Spandex a.k.a. Lycra a.k.a. Elastane: As you can guess from the name, it’s elastic, able to stretch up to 600x its original length. This is thanks to its structure with polymer chains that are both flexible enough to stretch, but also rigid enough to maintain its shape. The material is light, comfy, and bacteria-resistant thanks to treatments done in the manufacturing process, and UV-resistant thanks to the addition of UV inhibitors in the process of making it, as well as dries quickly, hence, it’s used to make swimwear as well as crepe bandages.
However, the fibres are prone to UV degradation, and since it stretches, it creates microscopic gaps that may allow UV to enter and affect the body.
Gore-tex (it’s not what it sounds like, I swear)
This is a material where a thin sheet of plastic Teflon is stretched between two layers of fabric. The sheet itself has holes big enough to allow sweat droplets to escape, but doesn’t allow water droplets from the outside to enter, keeping out the rain.
Teflon itself is made of the monomer tetrafluoroethene (or tetrafluoroethylene, if you prefer). Gore-tex is mainly used in waterproof clothing and shoes.
Thinsulate
This material is made of fine polypropene fibres about 1/100th the thickness of human hair! Despite this, these fibres are arranged so as to trap air, a poor conductor of heat and therefore a good insulator, between them, to keep people warm. Furthermore, it’s a poor absorber of water, so you’re still warm even when you’re wet, making it flexible and a good choice for skiers.
Non-Polymers
Carbon Fibres

Carbon Fibres: These bad boys found their way into sports equipment thanks to how strong they are despite their light weight. However, they aren’t a single material but rather two different materials, including thin, graphite-like carbon fibres mixed with plastic epoxy resin. It’s used in sports bikes, golf club shafts, and tennis rackets, as well as gym equipment too.
Titanium
Titanium: Despite being very strong and having low fatigue resistance, which is to say it doesn’t weaken over time, it is lightweight and has low density. Thus, it is used in golf clubs and scuba diving equipment, because it is also corrosion resistant, which is needed in the presence of salt water. Furthermore, as it doesn’t affect the human skin by not causing allergies, it is used in artificial hearts and bone implants.
Smart Materials
These bad boys change in response to changes in their environment, such as:
Chromogenic Materials
A change in voltage or temperature causes their colour to change. Examples include liquid crystal displays and light-sensitive sunglasses.
Non-Newtonian Fluids
Depending on the force applied, these liquids become more or less viscous. A famous example is Oobleck, a mixture of corn flour and water, which turns into a solid temporarily if you apply a large amount of force, and turns into a liquid if you apply less force.
Piezoelectric materials change shape when the voltage applied is changed.
Hydrogels
When the environment changes, they change their shape, too! They also have the ability to absorb 500 times its own weight in water, hence used in absorbent materials like baby napkins, water crystals, and wound dressings. This is because the negative ions in the hydrogel attract water molecules. They contain cross-linked carboxylic acids, which is to say, linear chains of carboxylic acids bond vertically with each other instead of horizontally, meaning molecules between chains can be eliminated vertically. You can find these in hair gels, plant water crystals, wound dressings, and disposable baby napkins.
However, if salt is added to the hydrogel, it will prefer to attract sodium ions instead of water, causing it to lose water. A similar effect occurs with changes in pH.
Shape Memory Alloys
Though bendable, if you heat these alloys, they return to their original shape. This is because they have two different crystal structures, one being stable at higher temperatures (heating) and the other stable at lower temperatures (room temperature). This means at colder temperatures, these alloys are soft and easy to bend, but when heated, they’re harder. Thus, they’re used to make artificial hip joints. Examples include nitinol, a nickel-titanium alloy.
Nanoparticles!
These particles have a size range between 0.01–100 nanometers, and as a result have a high surface area to volume ratio, making them effective catalysts. Their small size makes them efficient at trapping air, too, making them good insulators, and their small shape allows for the manufacturing of better cutting tools and drill bits, making the blade and edges sharper.
They are also used in aircraft wings because their small size prevents disasters like nanocracking from happening, and makes the wings more lightweight for the same strength.
These can be used for transport of drugs within the body, such as gold nanoshells that carry anti-cancer drugs that it releases on impact in the acid environment of a tumor cell.
Molecular Gastronomy
Fancy words for those really cool dishes you see served at posh restaurants which seem to defy the laws of science but also come in really small portions that cost way too much.
A good example of the use of chemistry in cooking is emulsions, as many dressings and sauces like mayonnaise are emulsions of oil and water, held together by emulsifiers like egg yolks.
Another example is foam, used in dishes like pavlova to make them airy, fluffy, and light, which are then baked for a crumbly texture. This is made from egg whites and water, with the proteins in the egg whites trapping air to form a mesh structure that holds together, creating a stable foam.


• A pavlova next to Pavlova Cookie from CRK.
Spherification
Spherification is also a pretty good example, where liquids like sodium alginate are mixed with sodium alginate before being dropped into cold calcium chloride solution, creating small balls of liquid that have a membrane holding them together on the outside but are liquid within. This includes things like popping boba!
Liquid Nitrogen
Liquid nitrogen is used for rapid freezing to make semi-frozen desserts without waiting a long time for it to actually become partially frozen. It can also be used for a cool, fog-like effect.
Molecular gastronomy also uses juxtaposition and will position a less flavorful food next to a more flavorful one to bring out and enhance their own tastes.
Errors in Experiments
Rule of Thumb: To increase accuracy, repeat the number of trials!
Temperature
When measuring the temperature change in a reaction, errors may occur as heat is lost to the surroundings. We can avoid this by insulating the equipment using a material that is a poor conductor of heat, like wood (such as in copper calorimeters), or aluminum foil to trap heat.
Getting Gassy!
Oxygen is the most soluble gas.
Carbon dioxide is dissolved in cold drinks to make carbonic acid, which is what creates that fizz.
To test for water, if you add dehydrated copper sulfate to water, it will go from white to blue. Or you can add it to dehydrated cobalt chloride and it will go from red to blue.